About the
Project
Sanofi Japan was facing a clear shift: traditional face-to-face engagement with Healthcare Providers (HCPs) was losing effectiveness. Cardiovascular specialists — particularly those managing acute coronary syndrome and PCI procedures—were increasingly difficult to access, with limited visibility into the drivers behind declining engagement.
Ekino was engaged to diagnose the root causes of this change—understanding how HCPs consume medical information, identifying friction within existing channels, and assessing the effectiveness of current approaches. The outcome established an evidence-based foundation for a more effective, scalable engagement model, positioned as a pilot to inform broader transformation across Sanofi’s portfolio.
-
ClientSanofi KK Japan
-
Year2019
-
RoleStrategy & Product Design Lead
-
AgencyEkino
Discovery
Workshop
We kicked off with a focused discovery workshop to align stakeholders and immerse teams in the problem space. Cross-functional teams from the Complavin brand, alongside control teams from Plaquenil and Allegra, were brought together to build design thinking capability and test whether insights could scale beyond the pilot.
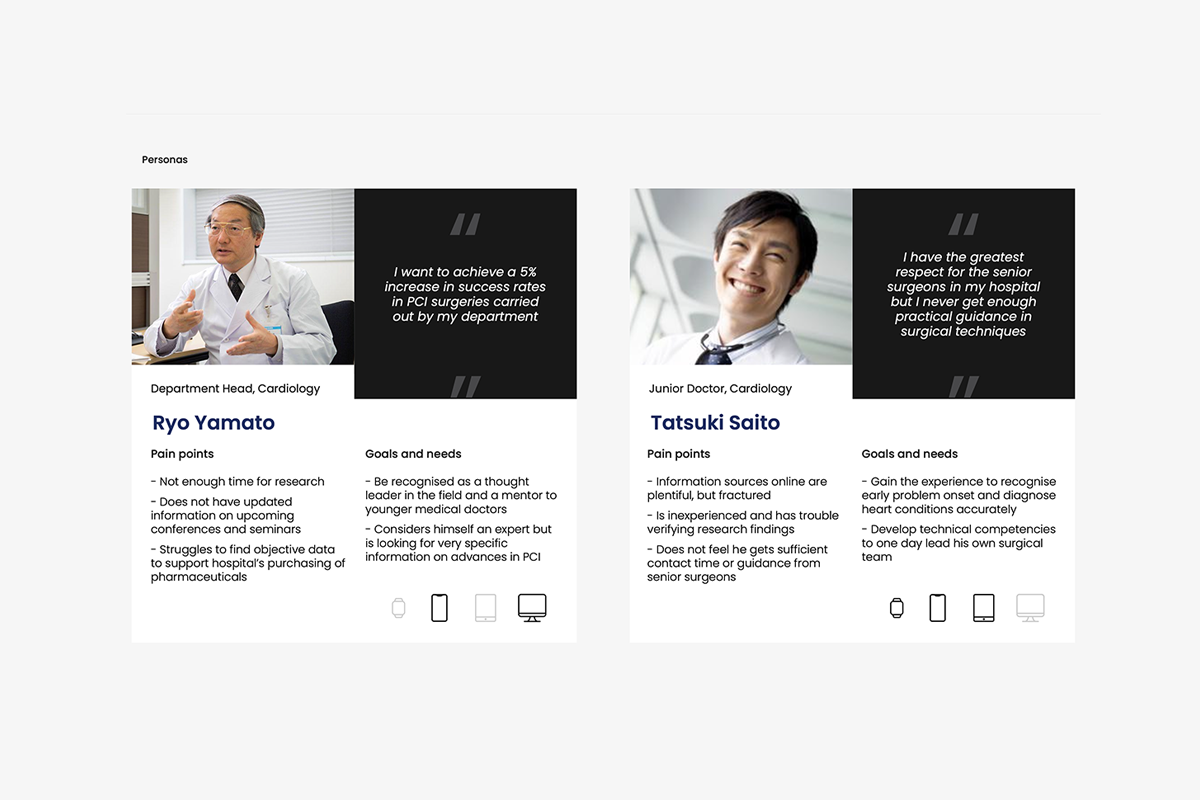
Through facilitated exercises, teams unpacked key HCP personas within the Japanese context, surfacing cultural and organisational dynamics shaping behaviour. “Day-in-the-life” mapping challenged assumptions around how HCPs manage time, access information, and engage with industry.
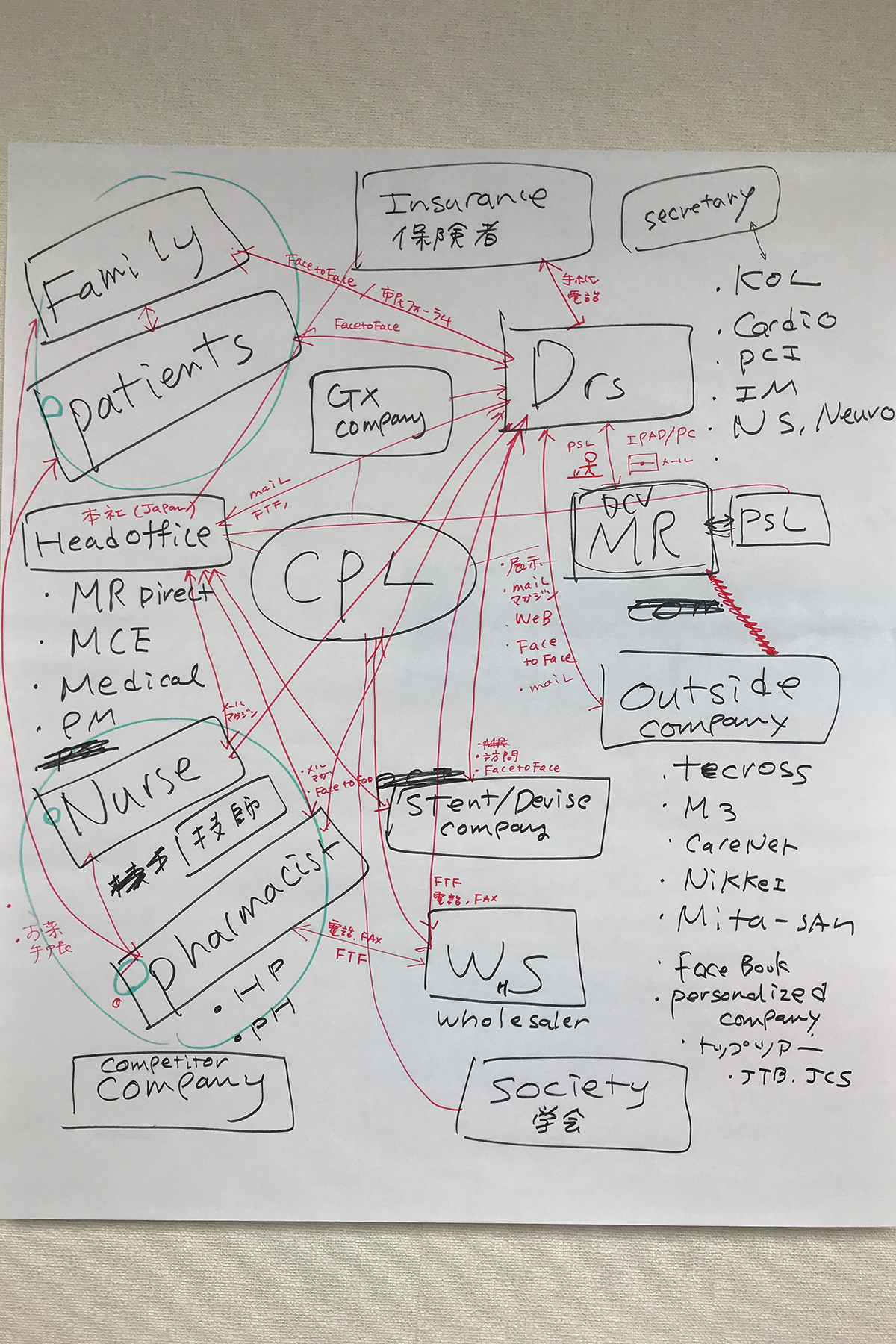
We also mapped the pharmaceutical and hospital ecosystem to identify key stakeholders, influence points, and structural constraints—clarifying where meaningful intervention was possible. This established a grounded, system-level view of the environment and its real-world challenges.
Validating
Assumptions
We conducted 24 in-depth interviews with cardiovascular HCPs across experience levels—from senior surgeons to junior practitioners—revealing distinct but equally critical needs. Senior surgeons were driven by reputation and recognition, while juniors sought broader learning through peer networks and hands-on exposure.
The core barrier was not time scarcity on the part of HCPs, but the active avoidance of pharma — driven by a perception that pharma prioritised product promotion over patient outcomes. Essentially, HCPs in Japan did NOT trust Big Pharma.
HCPs favoured credible, unbiased sources such as journals and clinical data, but access remained constrained, with high subscription costs leaving many with limited or no access to the information they actively sought.
Co-Creation
Workshop
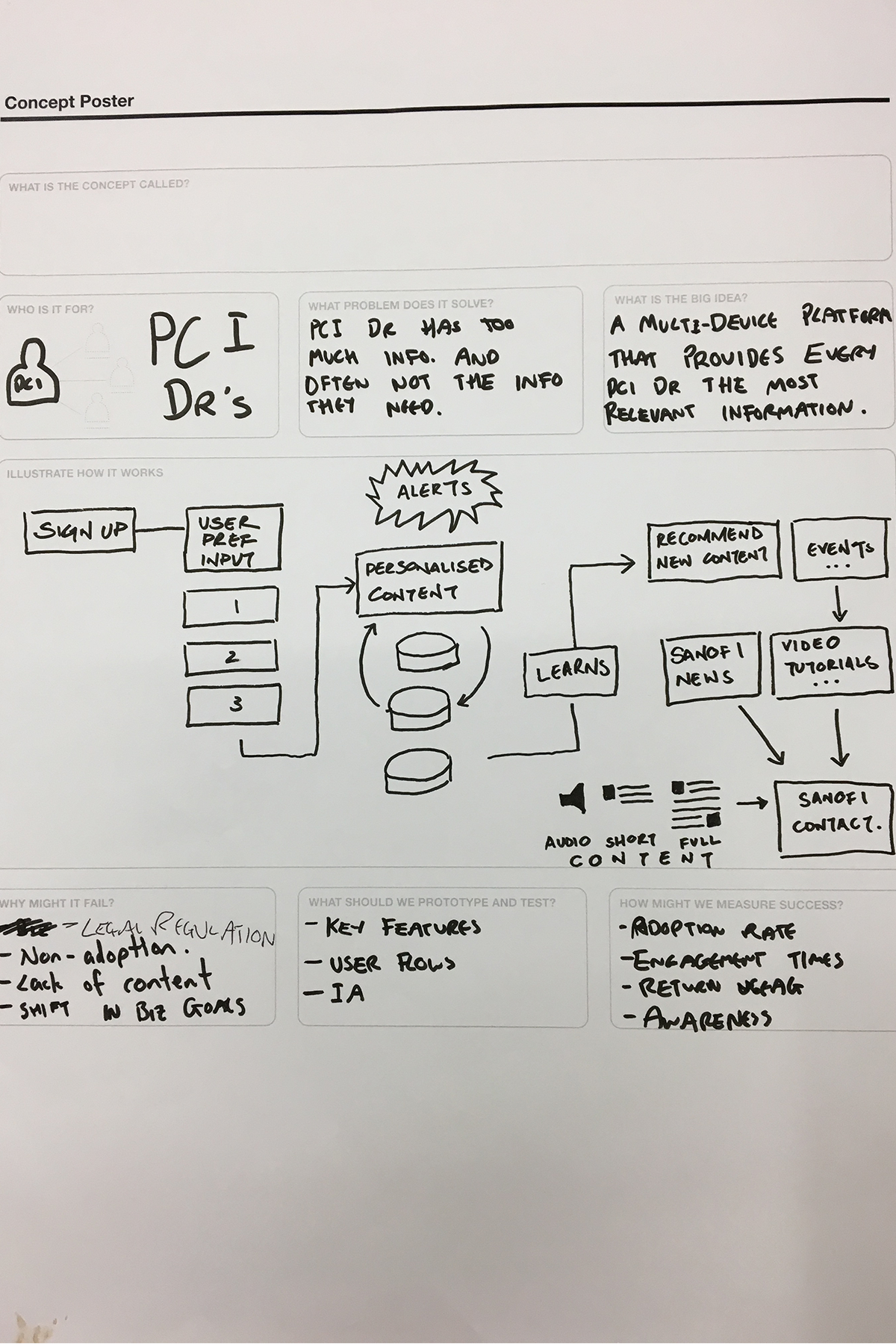
We reconvened teams to present and pressure-test the findings—most notably, the clear lack of trust in big pharma, driven by perceptions of product-led bias. Rather than working around this, we treated it as the core opportunity.
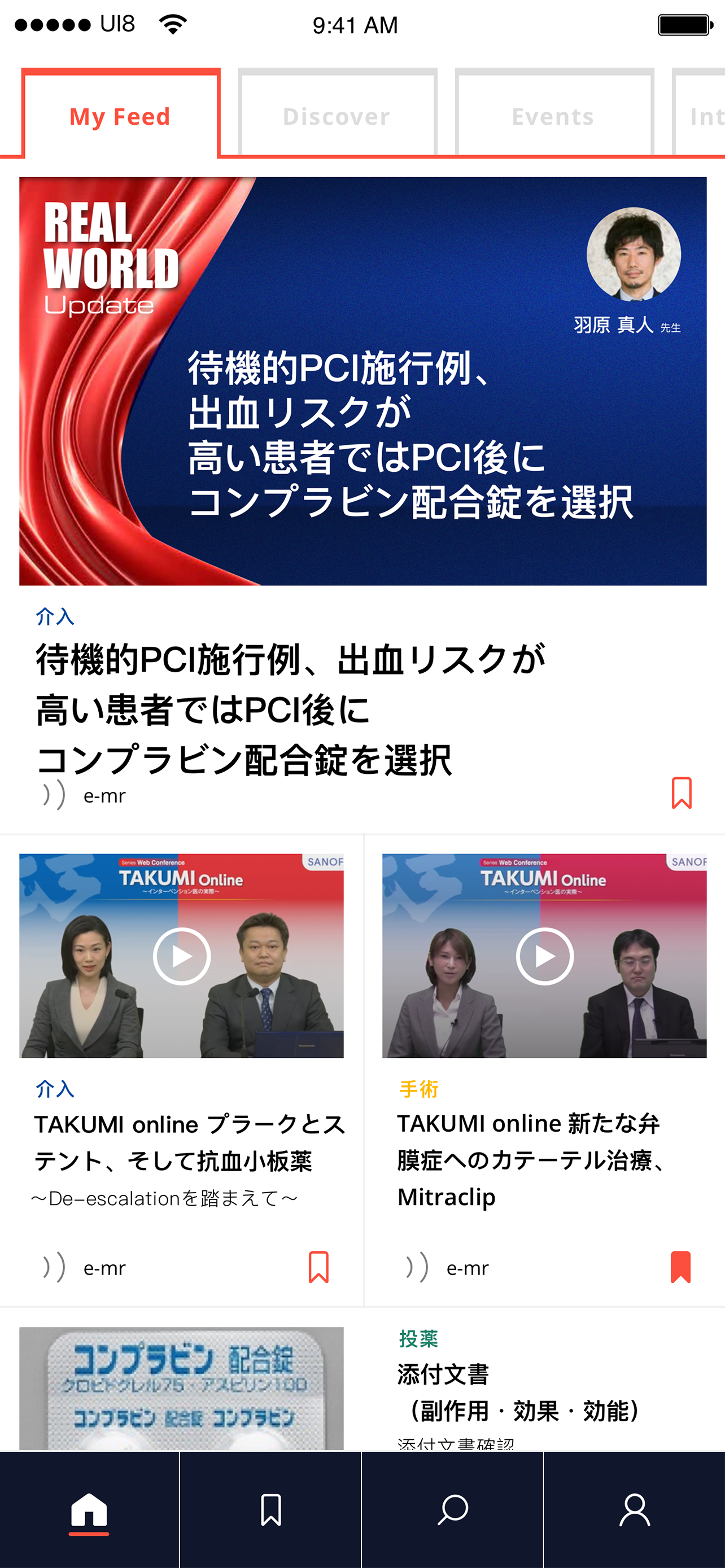
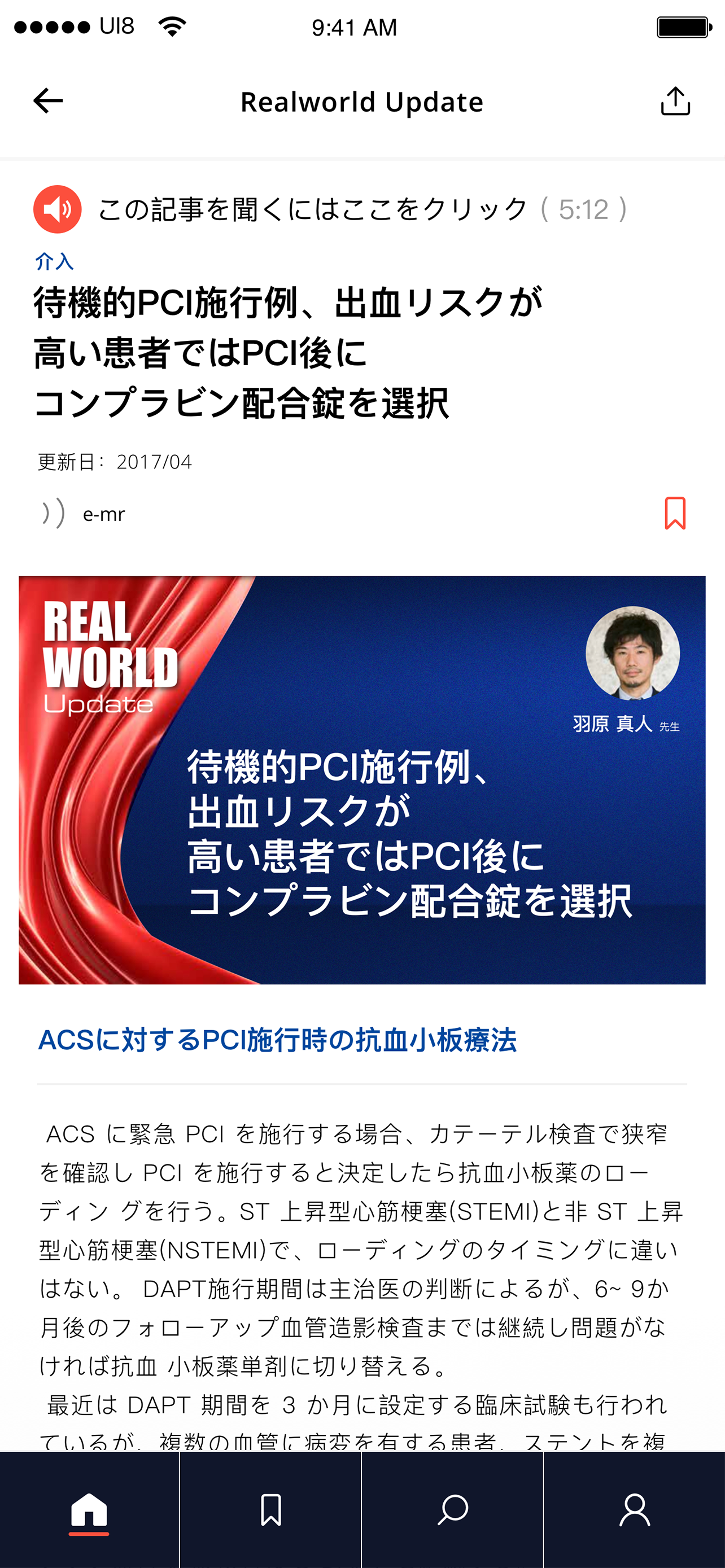
This led to a defining idea—our core service design innovation: a Sanofi-funded, unbranded platform providing access to credible, verified medical and scientific information. Built around real HCP behaviours, it delivered curated, specialty-relevant content, bite-sized summaries with deeper dive options, on-the-go access via text-to-speech, and peer learning through content sharing and expert-led sessions connecting senior and junior practitioners.
Rapid
Prototyping
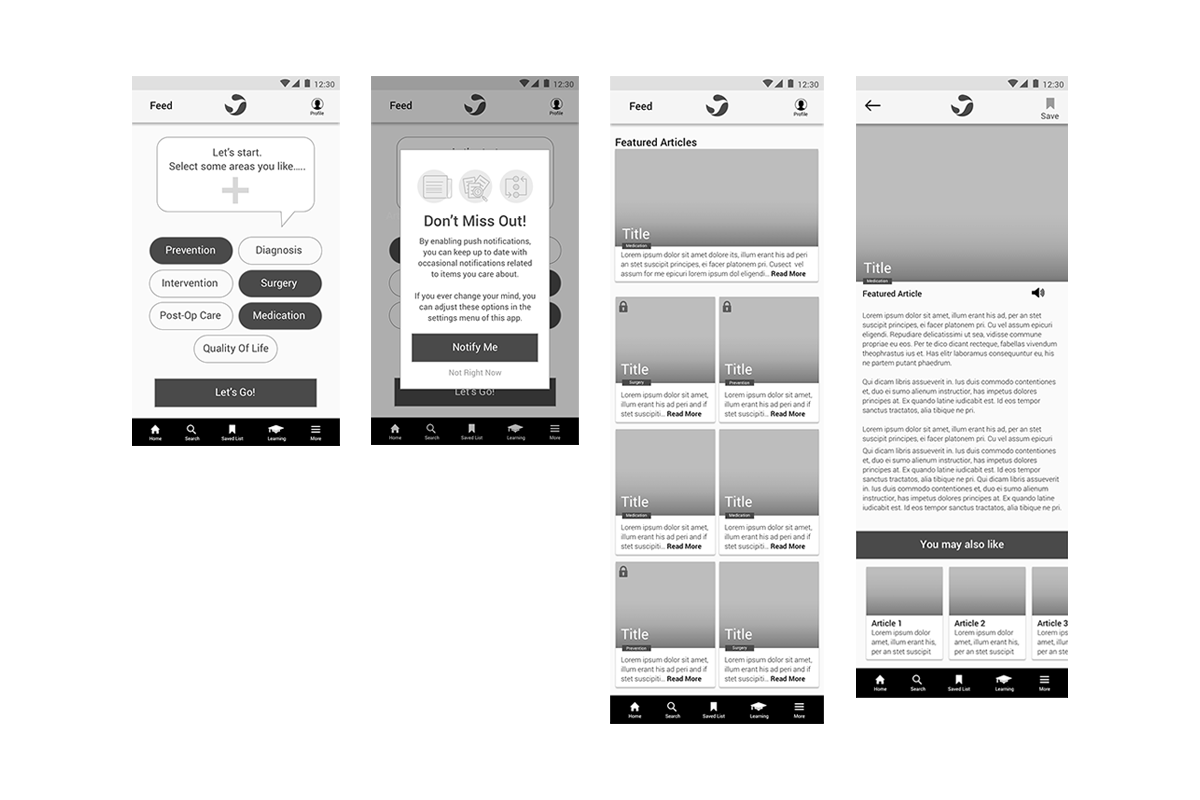
The design team moved at pace through a series of focused sprints, rapidly translating early concepts into lo-fi wireframes to validate core service flows and feature sets. This allowed us to test assumptions quickly, align stakeholders, and iterate in real time—ensuring the experience was grounded in actual HCP needs rather than internal hypotheses.
In parallel, we established a foundational design system to bring consistency, scalability, and speed to development. This enabled a seamless transition into a high-fidelity prototype that brought the service to life—showcasing key features, interaction patterns, and the overall user experience, while providing a tangible artefact to drive stakeholder buy-in and accelerate decision-making.
User Testing
& Iteration
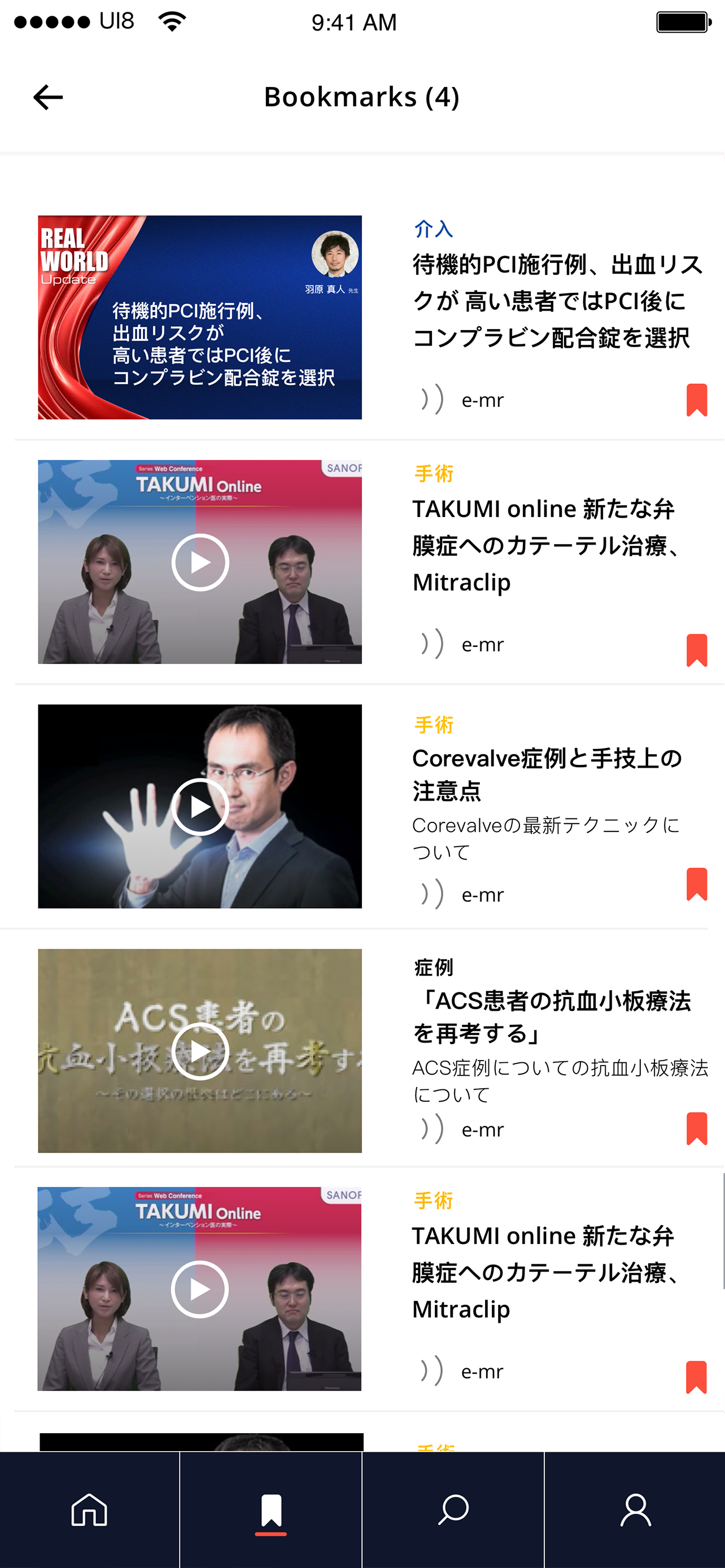
User testing was conducted in a controlled environment, with the service introduced as fully unbranded and not affiliated with any pharmaceutical company. HCPs responded positively, indicating strong intent to engage. They valued access to peer-reviewed articles, journals, and white papers, and appreciated features that reflected how they work—selecting areas of interest, diving deeper into content, saving for later, and consuming information on the go through text-to-audio..
The pivotal moment came when we revealed the service was funded by Sanofi. Rather than reinforcing scepticism, perceptions shifted. HCPs saw it as a meaningful contribution—supporting their development as clinicians and helping them deliver better patient outcomes. What began as a trust barrier became a lever for differentiation, reframing Sanofi’s role from product provider to enabler of better care.
Launching
The Pilot
The strength of user testing did more than validate the concept—it shifted internal conviction. What began as cautious exploration quickly turned into active sponsorship, with leadership recognising the potential to address a long-standing engagement gap. This marked a clear inflection point, moving the initiative from proof of concept into a funded pilot.
Feedback from HCPs was synthesised into targeted design iterations, sharpening both the experience and feature set. The result was a refined platform ready for real-world deployment—rolled out as a pilot to 700 doctors across Japan, signalling both confidence in the approach and a tangible step toward scaling a new model of HCP engagement.
Key
Impact
- Shifted HCP perception from scepticism to trust — repositioning Sanofi as a credible partner in clinical development, not just a product-driven pharma player
- +20% increase in HCP engagement, with stronger, more meaningful interactions between physicians and medical representatives
- 60% DAU/MAU ratio, signalling strong, sustained digital engagement and clear product-market fit for the platform
- Recognised at the Sanofi Global Innovation Summit 2018, showcasing the initiative as a benchmark for innovation within the global organisation