About the
Project
At Johnson & Johnson’s APAC Centre of Excellence, I led experience strategy for the regional launch of CARVYKTI—a pioneering cell and gene therapy for patients with Multiple Myeloma. Operating within a highly complex and regulated ecosystem, the work focused on building a deep, evidence-based understanding of the disease, while mapping end-to-end patient and HCP journeys across fragmented care pathways.
This foundation informed the design of an integrated HCP experience and service model—aligning medical, commercial, and operational stakeholders—to enable safe, scalable adoption across APAC markets and support the successful introduction of a first-in-class therapy.
-
CompanyJohnson & Johnson
-
Year2022
-
RoleStrategy & Innovation Lead
Shifting
Mindsets
Johnson & Johnson entered Multiple Myeloma as a market leader, driven by first-mover advantage and best-in-class therapies. In this model, success was product-led — efficacy was well-documented, pathways were established, and HCP confidence was high. These therapies were clinically effective in driving remission and their adoption was straightforward.
CAR-T therapies changed that.
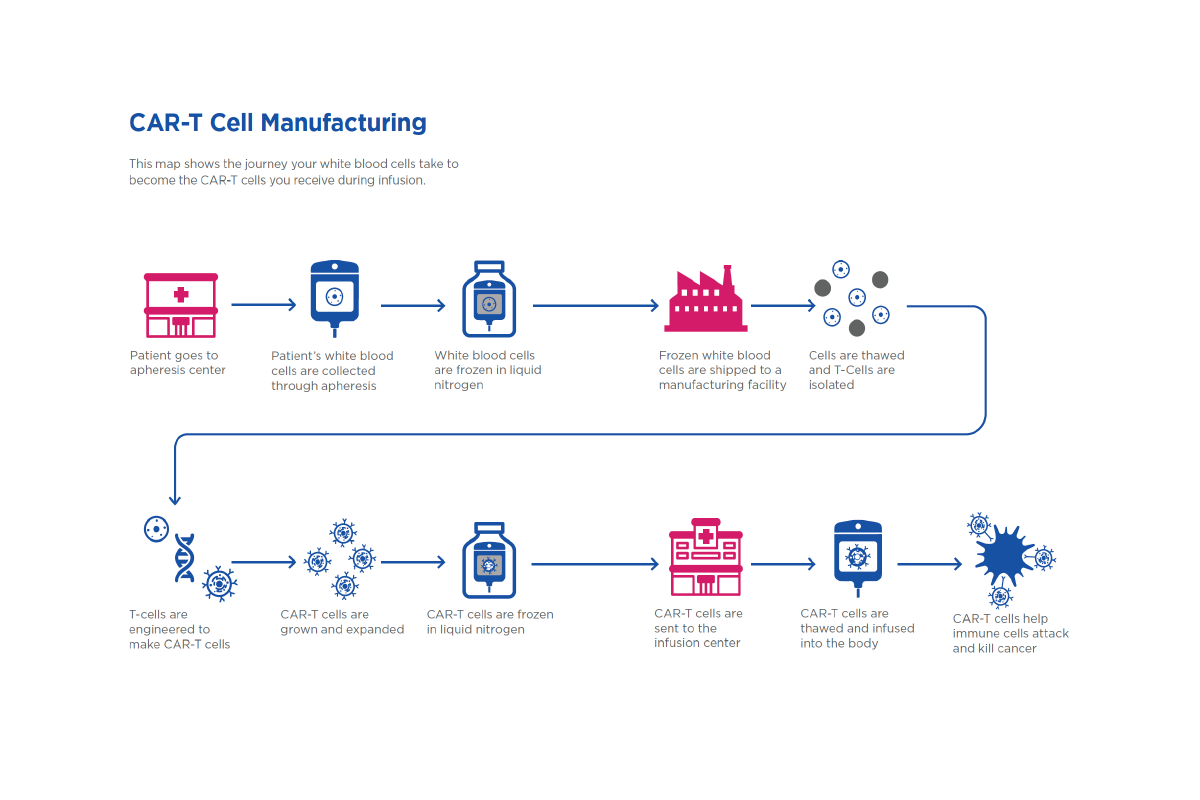
With CAR-T cell therapy, the potential for long-term remission — pussibly even a cure — emerged. But so did complexity. Treatment journeys extended up to 18 months, pathways became fragmented across stakeholders, HCP familiarity varied, and costs were significant. Clinical innovation alone was no longer enough to drive adoption.
The challenge shifted from product to experience.
Driving uptake required rethinking the entire ecosystem—moving beyond linear journeys to uncover real-world friction at every touchpoint. This meant deeply understanding both HCP and patient experiences, identifying micro-barriers, and designing targeted interventions to enable earlier decisions, build confidence, and coordinate care.
Aligning
Teams
Shifting to CAR-T required more than a new therapy—it demanded a reset in how the organisation thought. The starting point moved from product performance and business KPIs to patient outcomes and real-world experience, without losing sight of commercial impact. This shift was not trivial. Cell and gene therapy operates across a complex, globally distributed supply chain—an operational challenge intensified by COVID—making coordination, timing, and trust critical to success.
We began by bringing stakeholders together through a series of kickoff workshops. These sessions deliberately challenged our inherited assumptions—built on years of success in traditional therapies—and exposed their limitations in the context of a fundamentally different treatment paradigm. What worked before would not hold in a world where HCPs, patients, and caregivers faced significantly higher time, infrastructure, and cost commitments.
This phase created critical momentum. We aligned on a clear north star—defining the level of service and experience we aimed to deliver across key moments that matter. Teams acknowledged the existence of silos and blind spots, unlocking a shift toward systems thinking across functions and markets. Most importantly, there was a collective recognition that we did not yet understand the lived realities of our users deeply enough—creating the mandate, and securing investment, for ethnographic research to uncover the true barriers to adoption.
Moments
of truth
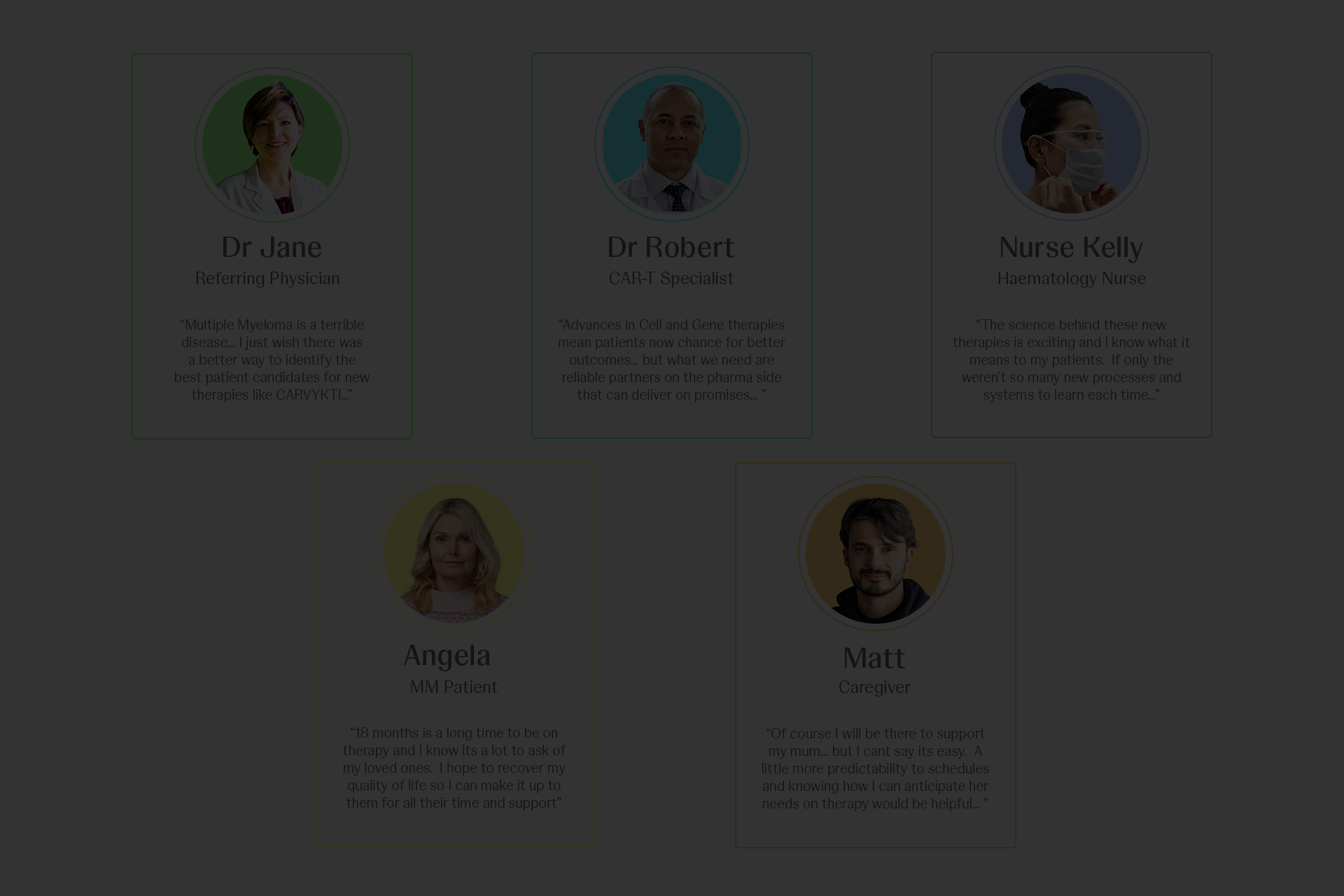
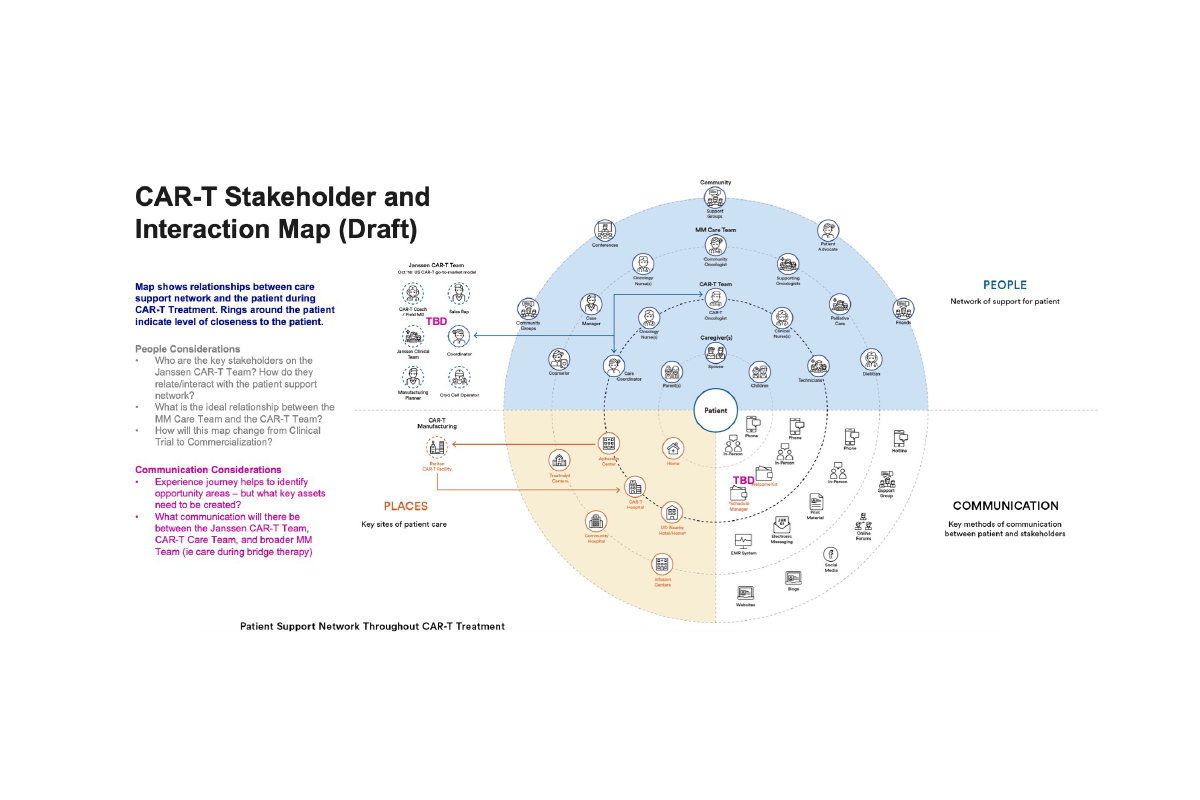
Research was led by an external agency to ensure objectivity and depth. We engaged directly with HCPs, nurses, patients, and caregivers to capture first-hand experiences, complemented by insights from nursing associations, patient support groups, and disease advocacy organisations. This multi-source approach gave us a grounded, end-to-end view of the ecosystem—beyond what internal data or assumptions could reveal.
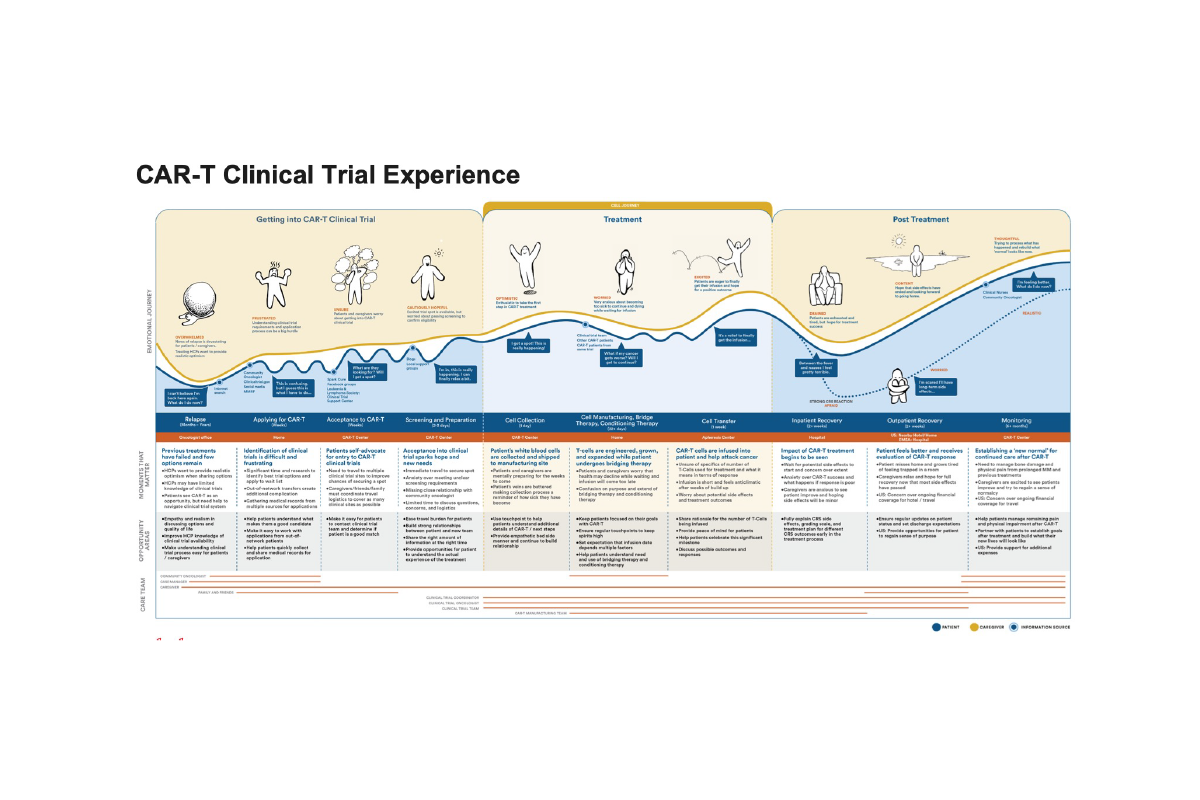
The findings were unequivocal. We uncovered friction across critical micro-moments in already complex journeys, alongside systemic gaps where services at treatment centres were often disjointed. When these insights were played back, the shift was immediate and visceral—teams connected emotionally with the reality of their users. That moment of clarity unlocked true alignment and buy-in, enabling us to move forward with conviction: to design and deliver a first-class, end-to-end service model that supports HCPs and nurses, improves patient outcomes, and meaningfully eases the burden on caregivers.

34 Specific steps
across 9 distinct phases
The Service
Blueprint
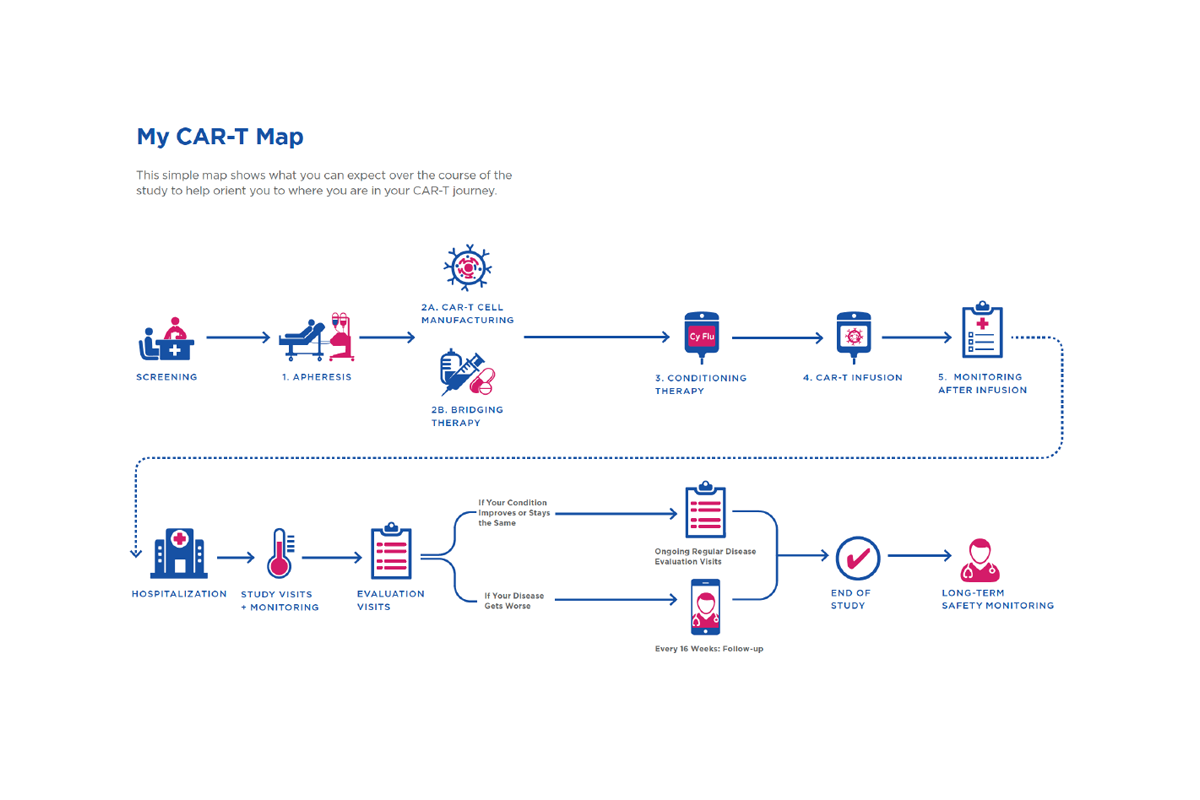
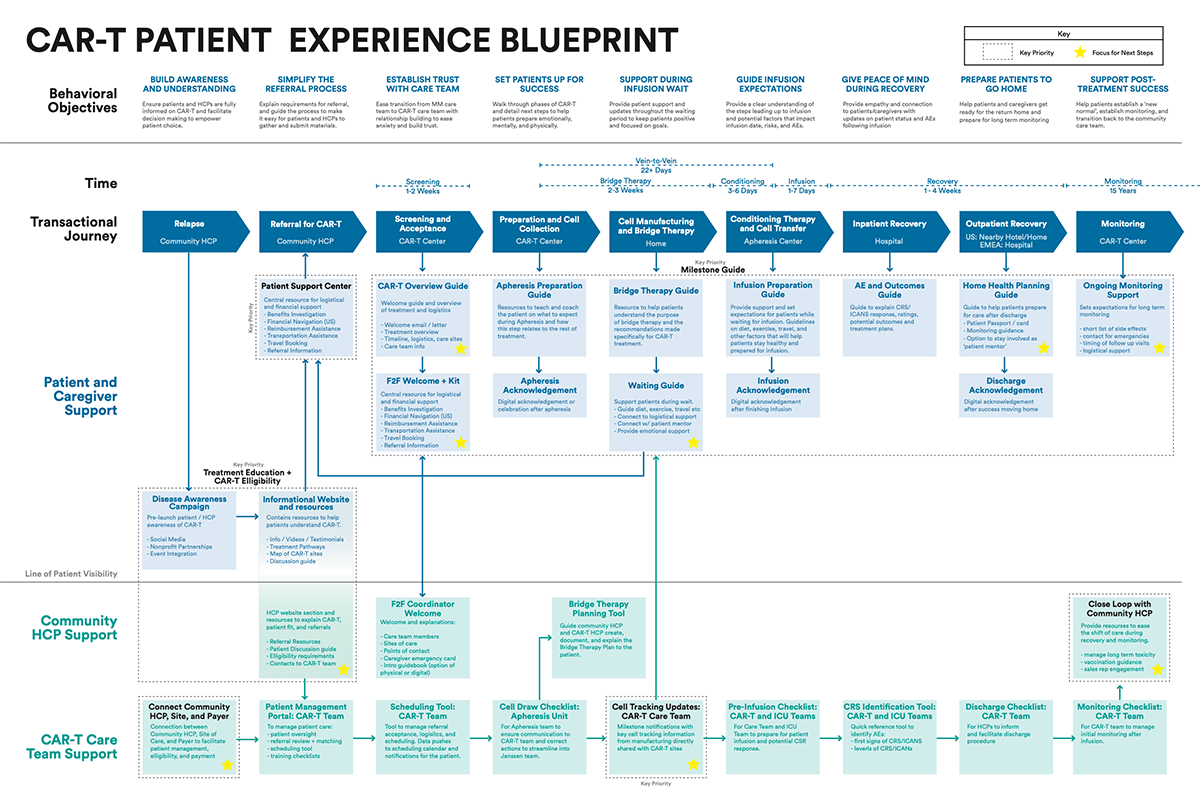
Building on the research, we redefined the journey from the ground up—expanding it from the original high-level 12-step view to a granular map of 34 distinct, intimate steps. This level of detail reflected the true complexity of CAR-T, capturing not just the clinical pathway, but the operational, emotional, and logistical realities experienced across the ecosystem.
At each step, we synthesised what was actually happening on the ground and defined the “jobs to be done” for every stakeholder involved—HCPs, nurses, patients, and caregivers. This created a shared, fact-based understanding of responsibilities, dependencies, and friction points, replacing assumptions with clarity.
Critically, we didn’t stop at mapping the current state. For every step, we defined what “good” should look like—articulating a clear, ideal experience from a Johnson & Johnson perspective. This established a consistent benchmark across markets and functions, turning the journey map into more than a diagnostic tool—it became a blueprint for designing interventions, aligning teams, and systematically elevating the end-to-end experience..
Strategic
Change
To operationalise the blueprint, we focused on three pillars: People, Solutions, and Processes—aligning mindsets and capabilities, enabling the right tools and services, and embedding governance and workflows to deliver a seamless, scalable experience.
People
Within the People pillar, we defined the human layer of the experience in detail—identifying, at each step, the front-stage interventions that would support HCPs, nurses, patients, and caregivers by removing friction or creating moments of value. These interventions were then mapped to specific roles within local market teams, establishing clear ownership across the journey. Beyond clarity, this approach enabled markets to right-size their team structures—adapting roles and resourcing based on market maturity, scale, and demand.
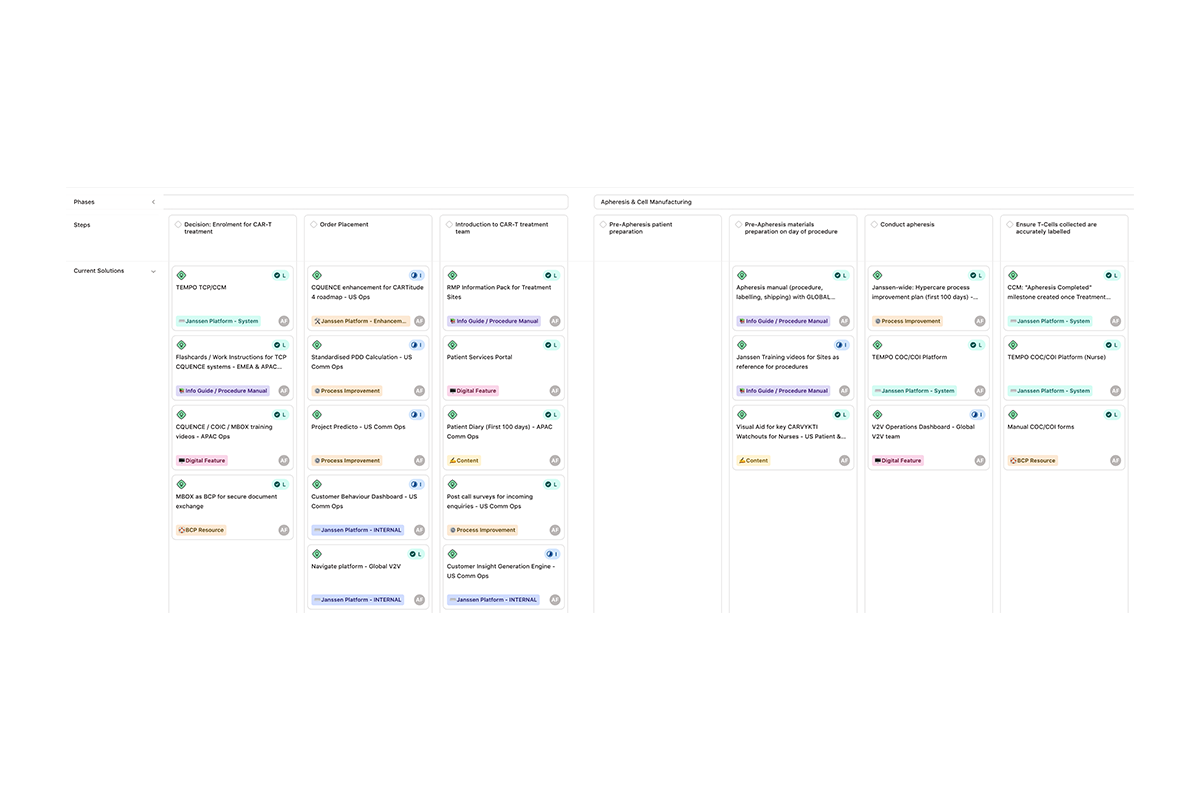
Solutions
Within the Solutions pillar, we assessed each step in the journey to identify what would meaningfully elevate the experience. This extended beyond an APAC lens—reviewing existing solutions across the US and EMEA to reuse, adapt, and avoid duplicative investment. At the same time, we identified new opportunities, evaluating them against viability, feasibility, and value creation. This created a structured decision-making framework, enabling markets to align on larger, regional investments while retaining flexibility to pursue smaller, market-specific solutions.
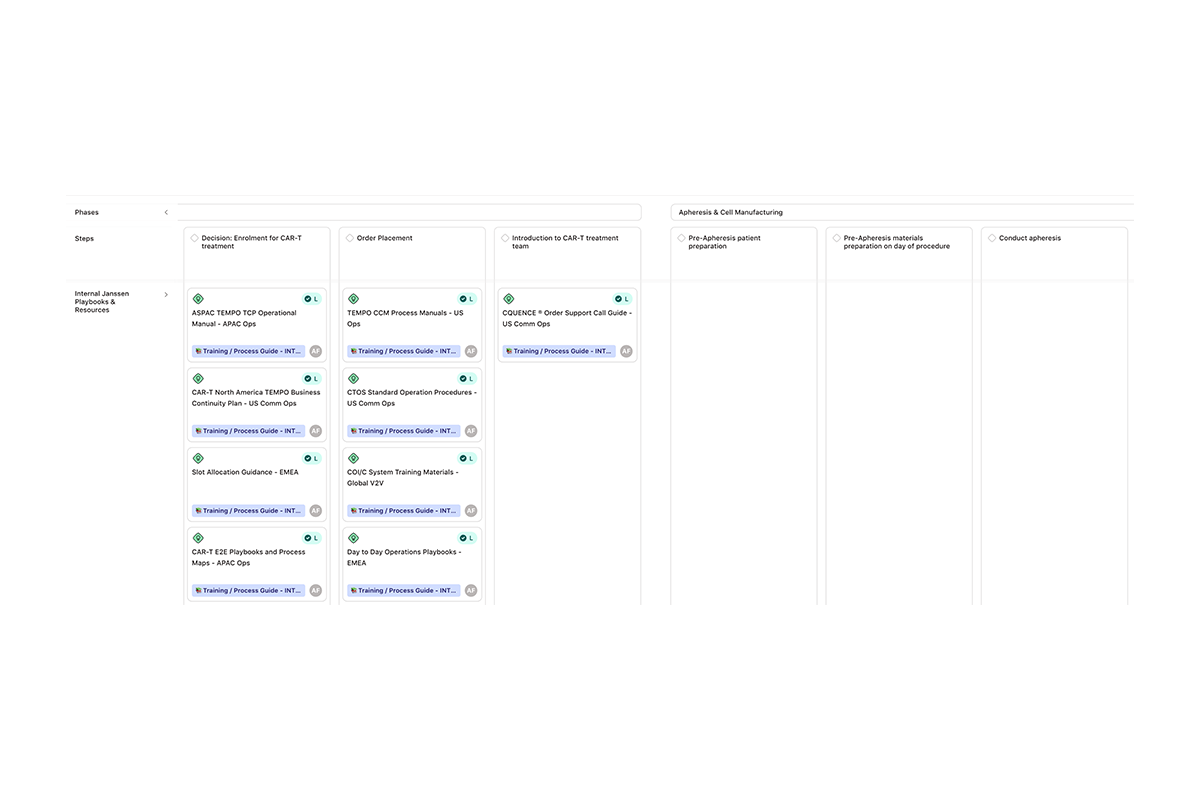
Processes
Within the Processes pillar, we focused on the backbone of delivery—redesigning both internal and external workflows and systems to reduce complexity and enable seamless coordination. For treatment centres and logistics partners, this meant simplifying interactions and aligning with familiar processes, removing the burden of learning entirely new system flows. Central to this was improving transparency and speed of communication, strengthening trust and credibility across the network.
Internally, we optimised channels and leveraged data to build targeted dashboards—equipping leadership and business units with timely, actionable insights to support faster, more informed decision-making.
Future
Proofing
To ensure sustained impact, the blueprint, as a living document, had to be supported by scalability, clear governance structures and ownership of artefacts, and regular review cadences. This ensured it remained current, actionable, and responsive to evolving market needs, while enabling consistent adoption and execution across markets.
Scalability
Designed initially for APAC—spanning the Centre of Excellence and five priority markets—the blueprint proved inherently scalable. Its clarity and utility drove strong reception when showcased in EMEA, leading to global adoption and the formation of a cross-regional working model to evolve it further. This expanded into a multi-tiered framework with varying levels of detail—global, regional, and local—each serving distinct planning and execution needs.
At the global level, it enabled horizon scanning and long-term investment planning. Regionally, it informed strategy, market expansion, and resource allocation. Locally, it translated into clear, executable actions. This structure also enabled the creation of a “Launch-in-a-Box” toolkit—allowing new markets to spin up quickly and consistently. With repeatable processes embedded and continuous updates from mature markets, the blueprint remained current—ensuring lessons learned were rapidly integrated and scaled across regions.
Governance
Governance was designed to ensure the blueprint remained active, relevant, and continuously improving. Clear ownership was established at every level — local market owners, regional leads, and global stewards — each accountable for maintaining and evolving their respective layers of the blueprint. A structured cadence supported this: local teams met biweekly to review progress, share learnings, and refine execution; regional leads convened monthly with markets to align on priorities, surface common challenges, and scale best practices; and global governance forums were held quarterly to consolidate insights, shape strategic direction, and guide investment decisions.
This rhythm created a two-way flow of intelligence — ground-level insights moved upward to inform strategy, while global priorities and innovations cascaded down into execution. Importantly, governance extended beyond meetings. It embedded clear decision rights, standardised artefact ownership, and version control—ensuring consistency while allowing for local adaptability. The result was a system that balanced alignment with agility, enabling the organisation to learn, adapt, and scale in real time.
Key
Impact
- Accelerated market adoption of a complex therapy — by removing friction across the end-to-end journey
- Reduced Go to Market timelines by 50% and training costs by 20%, through clarity in team roles, shape and a repeatable “Launch-in-a-Box” framework
- Moved the organisation from siloed execution to a coordinated, cross-functional system, — aligning Commercial, Medical, and Operations around a shared blueprint and common goals.
- Improved transparency, coordination, and support across treatment centres and patients, strengthening trust in both the therapy and J& J as a partner.
- Introduced dashboards and feedback loops that gave leadership real-time visibility supporting faster, more informed strategic and operational decisions.